
Summary
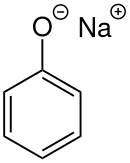
Sodium phenoxide (sodium phenolate) is an organic compound with the formula NaOC6H5. It is a white crystalline solid. Its anion, phenoxide, also known as phenolate, is the conjugate base of phenol. It is used as a precursor to many other organic compounds, such as aryl ethers.
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium phenoxide[1] | |
| Other names
Sodium phenolate
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider |
|
| ECHA InfoCard | 100.004.862 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H5NaO | |
| Molar mass | 116.09 g/mol |
| Appearance | White solid |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Harmful, Corrosive |
| Flash point | Non-flammable |
| Non-flammable | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Synthesis and structure edit
Most commonly, solutions of sodium phenoxide are produced by treating phenol with sodium hydroxide.[2] Anhydrous derivatives can be prepared by combining phenol and sodium. A related, updated procedure uses sodium methoxide instead of sodium hydroxide:[3]
- NaOCH3 + HOC6H5 → NaOC6H5 + HOCH3
Sodium phenoxide can also be produced by the "alkaline fusion" of benzenesulfonic acid, whereby the sulfonate groups are displaced by hydroxide:
- C6H5SO3Na + 2 NaOH → C6H5OH + Na2SO3
This route once was the principal industrial route to phenol.[citation needed]
Structure edit
Like other sodium alkoxides, solid sodium phenoxide adopts a complex structure involving multiple Na-O bonds. Solvent-free material is polymeric, each Na center being bound to three oxygen ligands as well as the phenyl ring. Adducts of sodium phenoxide are molecular, such as the cubane-type cluster [NaOPh]4(HMPA)4.[4]
Reactions edit
Sodium phenoxide is a moderately ?strong ?base. Acidification gives phenol:[5]
- PhOH ⇌ PhO− + H+ (K = 10−10)
The acid-base behavior is complicated by homoassociation, reflecting the association of phenol and phenoxide.[6]
Sodium phenoxide reacts with alkylating agents, such as trimethylene bromide, to afford alkyl phenyl ethers:[2]
- NaOC6H5 + RBr → ROC6H5 + NaBr
The conversion is an extension of the Williamson ether synthesis. With acylating agents, one obtains phenyl esters:[citation needed]
- NaOC6H5 + RC(O)Cl → RCO2C6H5 + NaCl
Sodium phenoxide is susceptible to certain types of electrophilic aromatic substitutions. For example, it reacts with carbon dioxide to form 2-hydroxybenzoate, the conjugate base of salicylic acid. In general however, electrophiles irreversibly attack the oxygen center in phenoxide.[citation needed]
References edit
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. pp. 1071, 1129. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ a b C. S. Marvel; A. L. Tanenbaum (1929). "γ-Phenoxypropyl Bromide". Org. Synth. 9: 72. doi:10.15227/orgsyn.009.0072.
- ^ Kornblum, Nathan; Lurie, Arnold P. (1959). "Heterogeneity as a Factor in the Alkylation of Ambident Anions: Phenoxide Ions1,2". Journal of the American Chemical Society. 81 (11): 2705–2715. doi:10.1021/ja01520a030.
- ^ Michael Kunert, Eckhard Dinjus, Maria Nauck, Joachim Sieler "Structure and Reactivity of Sodium Phenoxide - Following the Course of the Kolbe-Schmitt Reaction" Chemische Berichte 1997 Volume 130, Issue 10, pages 1461–1465. doi:10.1002/cber.19971301017
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 978-0-471-72091-1
- ^ K. Izutsu (1990). Acid-Base Dissociation Constants in Dipolar Aprotic Solvents. Vol. 35. Blackwell Scientific Publications.
External links edit
Media related to Sodium phenoxide at Wikimedia Commons


